Imagine unlocking the secret behind some of the most effective skincare products on the market. You’re about to discover Hydroxyethylpiperazine Ethane Sulfonic Acid, an ingredient that might be the missing piece in your skincare routine.
Have you ever wondered why certain products seem to work wonders while others fall short? This acid could be the answer. By understanding its role and benefits, you can make informed choices and elevate your skincare game. Stay with us as we dive deep into the world of Hydroxyethylpiperazine Ethane Sulfonic Acid and uncover how it can transform your skin’s health and appearance.
Whether you’re a skincare enthusiast or just looking to improve your current routine, this information is tailored for you.
Chemical Composition
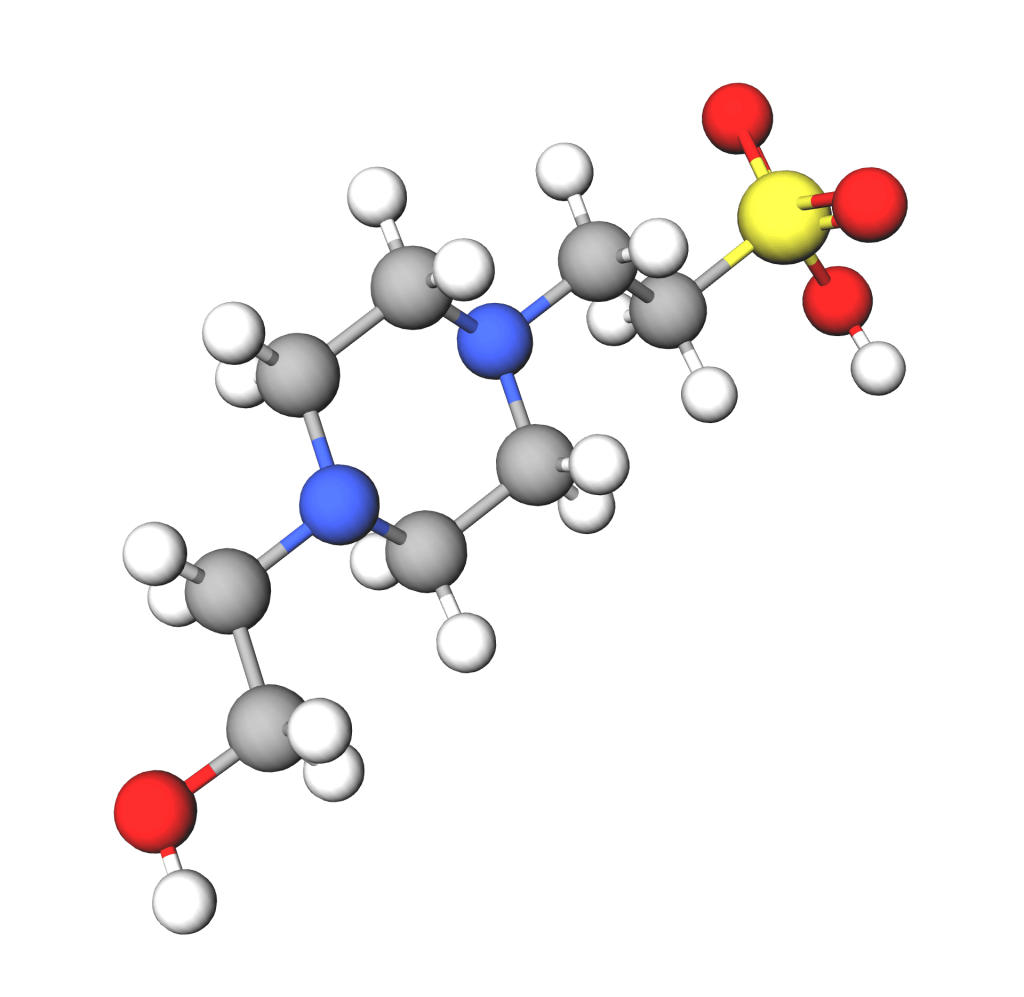
Understanding the chemical composition of Hydroxyethylpiperazine Ethane Sulfonic Acid might seem complex, but it’s actually quite fascinating. This ingredient, often referred to as HEPES, plays a crucial role in various applications. Whether you’re a science enthusiast or someone curious about the components in skincare products, knowing how HEPES is structured can help you appreciate its benefits.
Structure And Composition
HEPES is a zwitterionic buffer, which means it has both positive and negative charges. This unique structure allows it to maintain a stable pH in solutions. Imagine a seesaw, perfectly balanced; that’s how HEPES works to keep things steady.
Its molecular formula is C8H18N2O4S. With eight carbon atoms, it provides a backbone that holds everything together. The nitrogen atoms contribute to its buffering capabilities, making it essential in biological environments.
Role In Stability
HEPES is known for its ability to stabilize pH levels. It’s like a guardian keeping the balance in various solutions. This stability is crucial in laboratories where precise conditions are needed for experiments.
Have you ever used a skincare product that felt just right on your skin? That’s often thanks to buffers like HEPES ensuring the product doesn’t irritate your skin by being too acidic or basic.
Applications In Daily Life
HEPES isn’t just a lab superstar; it has practical applications too. It’s found in many skincare products, helping to ensure they remain effective and gentle. Next time you check your skincare ingredients, look for HEPES.
It’s also used in cell cultures, ensuring that the environment remains conducive for growth. If you’ve ever wondered how scientists grow cells in the lab, buffers like HEPES are part of the magic.
Why Should You Care?
You might wonder why knowing about HEPES matters. Well, understanding ingredients can empower you to make informed choices about products you use daily. Plus, knowing about HEPES can spark conversations that lead to new insights.
Consider this: What other products might benefit from the stability HEPES offers? Could it improve the reliability of something you use regularly? Asking these questions can inspire innovative ideas and solutions.
Role In Buffer Solutions
Hydroxyethylpiperazine ethane sulfonic acid, often called HEPES, plays a vital role in buffer solutions. Buffers maintain stable pH levels, crucial for biological and chemical processes. HEPES stands out for its ability to keep pH constant, even with temperature changes or dilution.
Understanding Buffer Solutions
Buffer solutions resist pH changes. They consist of a weak acid and its salt or a weak base and its salt. This combination helps absorb excess H+ or OH- ions. Buffers are essential in labs, research, and industries.
Hepes In Buffer Formulation
HEPES is a popular choice in buffer formulation. It offers a stable pH range from 6.8 to 8.2. This range covers many biological applications. HEPES is effective in cell culture, enzyme studies, and protein research.
Advantages Over Other Buffers
HEPES is more stable than bicarbonate buffers. It does not react with metal ions. This makes it suitable for sensitive experiments. Its low ionic strength is ideal for electrophoresis and chromatography.
Application In Biological Research
HEPES is widely used in biological research. It supports cell viability in culture media. Researchers trust it for accurate pH control in experiments. Its non-toxic nature ensures safe interaction with cells.
Environmental Stability
HEPES buffers are environmentally stable. They withstand temperature fluctuations. This stability is crucial for outdoor or field experiments. HEPES remains reliable in varied environmental conditions.
Applications In Pharmaceuticals
Hydroxyethylpiperazine ethane sulfonic acid (HEPES) plays a crucial role in pharmaceuticals. This organic compound serves as a buffer agent. It maintains the stability of drug formulations. It ensures drugs remain effective and safe for use. Let’s explore how HEPES is applied in pharmaceuticals.
Applications In Drug Formulations
HEPES helps maintain pH balance in drug formulations. It prevents degradation of active ingredients. This ensures medicines are effective throughout their shelf life. Many liquid pharmaceuticals rely on HEPES for stability.
Role In Injectable Solutions
HEPES is often used in injectable solutions. It provides a stable pH environment. This stability enhances patient safety. Patients receive consistent and effective treatment with these solutions.
Hepes In Vaccines
Many vaccines contain HEPES as a buffering agent. It maintains the vaccine’s stability and effectiveness. This ensures vaccines remain potent during storage and transportation. It is a vital component in vaccine production.
Use In Biotechnology Products
Biotechnology products often use HEPES. It helps in maintaining cell culture environments. This is critical for producing consistent and reliable biotechnological products. It ensures cell health and viability.
Hepes In Ophthalmic Solutions
HEPES is used in ophthalmic solutions. It prevents irritation and maintains pH balance. Patients experience comfort and effective treatment. It is a key component in eye care products.
Use In Laboratory Research
Hydroxyethylpiperazine Ethane Sulfonic Acid, commonly known as HEPES, plays a crucial role in laboratory research. This organic chemical buffer is a staple in many labs due to its ability to maintain pH levels in cell culture media. Its stability makes it an essential ingredient for a wide range of scientific experiments.
Hepes In Cell Culture
In cell culture, maintaining a stable pH is critical for cell viability and function. HEPES provides this stability, even when there are changes in carbon dioxide levels. This ensures that your experiments yield reliable and consistent results.
Have you ever had cell culture experiments fail due to pH fluctuations? This is where HEPES shines, providing you peace of mind and saving time.
Role In Protein Studies
When studying proteins, maintaining a specific environment is essential. HEPES is often used in protein purification and crystallization processes. Its buffering capacity ensures that proteins maintain their structure and function throughout the experiment.
Imagine running an experiment where the protein denatures because of an unsuitable buffer. HEPES prevents such mishaps, keeping your research on track.
Compatibility With Biological Samples
HEPES is compatible with a variety of biological samples. This makes it versatile for different types of research, from microbiology to molecular biology. Its non-toxic nature ensures that it won’t interfere with biological reactions.
When choosing a buffer for your experiments, do you consider its impact on your samples? HEPES minimizes these concerns, allowing you to focus on your research outcomes.
Considerations For Use
While HEPES is a powerful buffer, it’s essential to use it correctly. Ensure the concentration is appropriate for your specific experiment. Also, consider its interaction with other reagents to avoid unexpected results.
Have you checked if your buffer is affecting your results? Taking the time to optimize HEPES usage can make a significant difference in your research success.
Safety And Handling
Hydroxyethylpiperazine ethane sulfonic acid requires careful handling to ensure safety. Always wear protective gear, like gloves and goggles, when working with it. Store in a cool, dry place, away from incompatible substances.
When using chemicals like Hydroxyethylpiperazine Ethane Sulfonic Acid (HEPES), understanding safety and handling is crucial. Whether you work in a lab or simply have a curious mind, knowing how to handle HEPES safely can prevent accidents and ensure a smooth experience. Here, we delve into practical tips and guidelines for managing this chemical responsibly.Understanding Safety Labels
Safety labels on HEPES containers are your first line of defense. These labels provide essential information about potential hazards. Always read them before opening a container to ensure you know how to handle the substance safely.Proper Storage Practices
Storing HEPES correctly can prolong its shelf life and maintain safety. Keep it in a cool, dry place away from direct sunlight. Ensure that it’s stored in a well-ventilated area to prevent any buildup of fumes.Personal Protective Equipment (ppe)
Wearing the right personal protective equipment is key. Gloves, goggles, and lab coats are non-negotiable. This gear protects your skin and eyes from accidental spills and splashes.Handling Spills And Accidents
Accidents can happen, even with the best precautions. Knowing how to manage spills is essential. Have a spill kit on hand, and ensure you understand the cleanup procedure before starting your work.Disposal Procedures
Disposing of HEPES must be done carefully to prevent environmental harm. Follow your local regulations for chemical disposal. Never pour it down the sink or throw it in the trash.Health And First Aid Measures
Familiarize yourself with first aid measures in case of exposure. If HEPES comes into contact with skin, wash immediately with soap and water. For eye contact, rinse thoroughly and seek medical attention if irritation persists.Training And Education
Continuous learning is vital. Attend workshops or training sessions on chemical safety. Being informed not only protects you but also those around you. Have you ever thought about how a small oversight in safety can lead to big consequences? Taking the time to understand and implement these safety measures can make all the difference. Prioritize safety, and you’ll find that working with chemicals like HEPES becomes a much less daunting task.
Credit: www.ewg.org
Environmental Impact
Hydroxyethylpiperazine ethane sulfonic acid, often used in labs, has a minimal environmental footprint. Its stable nature reduces risks of environmental harm, making it safer for ecosystems. This chemical’s low reactivity helps limit pollution.
Environmental Impact of Hydroxyethylpiperazine Ethane Sulfonic Acid When it comes to understanding the environmental impact of chemicals, Hydroxyethylpiperazine Ethane Sulfonic Acid (HEPES) provides some interesting insights. As a widely used buffering agent in biological and chemical research, it’s crucial to consider how it affects our environment. Let’s dive into what makes HEPES stand out and what you should know.Production And Biodegradability
HEPES is synthesized in labs, and its production process is designed to be efficient. However, the question arises—how biodegradable is it? Research shows that HEPES is not readily biodegradable, which means it can persist in the environment for some time. While it doesn’t degrade quickly, it’s not deemed highly toxic to aquatic life. This balance is crucial. It suggests that HEPES may not immediately harm ecosystems, but its accumulation could pose long-term challenges.Impact On Water Systems
Once HEPES enters water systems, it can remain stable. This stability is beneficial for controlled experiments in labs but raises concerns in natural water bodies. Persistent chemicals in water can disrupt natural ecosystems over time. Have you ever wondered how your actions in the lab might affect the water you drink? This is why considering the disposal methods of lab chemicals is vital for environmental protection.Responsible Disposal Practices
Proper disposal of HEPES is essential to minimize its environmental footprint. Instead of pouring it down the drain, consider using designated chemical waste disposal services. This practice ensures that HEPES is handled in a way that prevents it from entering the water supply. Have you thought about what happens to chemicals after they leave the lab? By being mindful of disposal, you contribute to a healthier environment and demonstrate responsible research practices.Future Considerations And Alternatives
As awareness grows about the environmental impact of chemicals, researchers are exploring alternatives. Could there be more eco-friendly buffering agents on the horizon? The potential for innovation in this area is exciting. You might be part of the change by advocating for and experimenting with greener alternatives. This not only benefits the planet but also aligns with the growing trend towards sustainable science. By considering the environmental impact of HEPES, you take a step towards more responsible research practices. Every small action counts, and your choices can lead to meaningful change.Alternatives And Comparisons
Hydroxyethylpiperazine Ethane Sulfonic Acid (HEPES) is known for its buffering capabilities in laboratories. But what if you’re curious about alternatives? This section will explore a few options and their comparisons to HEPES. Understanding these alternatives helps make informed decisions for specific applications.
Comparison With Tris Buffer
Tris Buffer is another popular choice in labs. It offers effective buffering in biological applications. Unlike HEPES, Tris Buffer can influence experimental results due to its reactivity. Some researchers prefer HEPES for its stability and minimal interference.
Phosphate Buffered Saline (pbs)
PBS is common in cell culture and molecular biology. It is easy to prepare and affordable. While HEPES provides a more stable pH, PBS is still reliable for many tasks. Some experiments may benefit more from HEPES’s consistent environment.
Mes Buffer
MES Buffer is often used in biochemistry. It offers a similar pH range to HEPES. However, MES can be less effective in certain temperature ranges. HEPES’s versatility makes it useful in various settings. Choosing between them depends on specific research needs.
Hepes Vs. Good’s Buffers
Good’s Buffers are designed for biological research. They include several buffering agents like MOPS and TES. HEPES remains favored for its low impact on biological systems. Good’s Buffers provide diverse options but may not match HEPES’s stability.

Credit: www.revoxb77.com

Credit: mansonbio.en.made-in-china.com
Frequently Asked Questions
What Is Hydroxyethylpiperazine Ethane Sulfonic Acid?
Hydroxyethylpiperazine Ethane Sulfonic Acid, commonly known as HEPES, is a buffering agent. It is widely used in biological and biochemical research. HEPES stabilizes pH levels in cell culture and experimental solutions, ensuring accurate results. Its effectiveness makes it a popular choice among researchers.
How Does Hepes Buffer Work?
HEPES buffer works by maintaining stable pH levels in solutions. It resists changes in pH, which is crucial for experiments. HEPES is effective over a wide pH range, making it versatile. This stability helps ensure reliable and consistent results in biological research.
Is Hepes Safe For Cell Culture?
HEPES is considered safe for cell culture applications. It does not interfere with cellular processes and maintains pH stability. Researchers use it widely due to its non-toxic nature. However, it’s important to follow guidelines for concentration and use.
Why Choose Hepes Over Other Buffers?
HEPES offers superior pH stability compared to other buffers. Its effectiveness across a broad pH range is unmatched. It does not interfere with biological reactions. This makes HEPES a preferred choice for precise experimental conditions.
Conclusion
Hydroxyethylpiperazine ethane sulfonic acid plays a crucial role in science. It helps maintain stable pH levels in various solutions. This stability supports accurate experiments and reliable results. Researchers value its effectiveness and consistency. Its usage spans across numerous scientific fields.
From biology to chemistry, its impact is evident. Understanding this compound enhances our scientific knowledge. It also aids in advancements in technology. As research continues, its importance may grow further. Learning about such ingredients enriches our comprehension of science. Stay curious and informed about these essential components.
Science thrives on knowledge and discovery.